Geoffrey Tyndall - Atmospheric Chemist
Spying on molecules in motion

Geoff Tyndall may seem like a typical scientist, if such a thing exists. "I work in a lab and wear a white coat sometimes," he laughs.
Yet his research is anything but predictable. Geoff runs a chemical kinetics laboratory full of high-tech instrumentation, including reaction chambers, lasers, spectrometers, and other air sampling devices. "Basically, we have different metal and glass containers that we put gases into to try to simulate reactions in the atmosphere. We measure the composition before and after, and try to figure out what changes occurred and why," he explains. Spectrometry is a key technique: "We use absorption of light to tell us what's happening inside the reactor, so we don't have to disturb the sample."
Geoff is a senior scientist in NCAR's Atmospheric Chemistry Division. "Being a physical chemist, I'm interested in the fundamental side of things—it's interesting to know how something reacts in the atmosphere, but also to know why it reacts that way," he says.
A dance of chemical reactions
Geoff and colleagues John Orlando and Yongxin Tang focus mainly on emissions from molecules containing carbon, called organic compounds, such as those from vegetation or fossil fuels. For example, NCAR scientists may bring air sample containers back from a field project in the Amazon rainforest filled with little-studied compounds. In the lab, Geoff uses lasers and other sampling devices to learn more about the compounds.
"John and I have been working for 15 years on organic mechanisms, building up a big data base of knowledge that we can refer to. The idea being that if we study enough of these compounds, we can eventually predict the behavior of the ones that are less amenable to study," Geoff explains.
The basic questions he explores concern how quickly gases react after they're released into the atmosphere and what the products are. "When something reacts, what does it make? Is it more toxic than the stuff that went into the atmosphere in the first place?" for example.
One of Geoff's recent projects investigates reactions of fluorine-containing compounds, which are used in stain-resistant products. "These compounds get into the atmosphere and some of the degradation products are acids, which are pretty long lived," he says. "A lot of those acids end up in the food chain in the Arctic. We're trying to trace the pathways by which they're formed and where they go."
The kinetics team works closely with colleagues to better represent atmospheric chemistry in computer models. Researchers also come to the lab for help calibrating instruments before heading off to field projects.
Geoff thrives on the intellectual stimulation at work. "As a physical chemist, I'm interested in quantifying thing, putting numbers on them—how fast does this go, and why is this reaction faster than that one?". He also enjoys the hands-on aspect of time in the lab, as well as interacting with colleagues and university visitors. His biggest challenge is time management, as he's often juggling multiple projects at once.
From construction kit to chemical building blocks
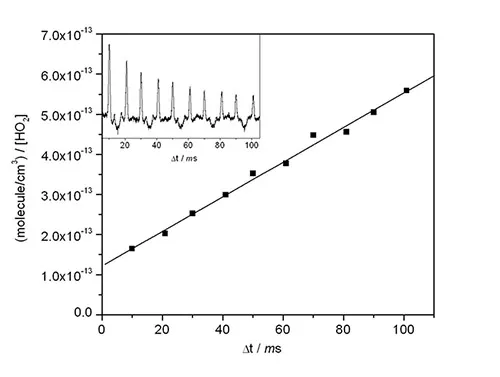
Hydroperoxy (HO2) radicals are central to the production of ozone and the generation of hydroxyl radicals through their interaction with nitrogen oxide. The NCAR kinetics group has developed a flash photolysis system with a solid-state diode laser to detect HO2 radicals. This figure shows the second-order decay of HO2 radicals. The diode laser is swept across an HO2 absorption feature to give a discrete measurement of HO2 every 10 milliseconds. The inset shows the HO2 absorptions, corresponding to an initial concentration of 8 x 1012 molecules per cubic centimeter.
Illustration courtesy Geoffrey Tyndall
Geoff grew up in England's coal-mining region just north of Nottingham, in the middle of what is left of Sherwood Forest. His father was an engineer in the coal industry. "We had a Meccano kit," a popular model construction kit, he recalls. "My dad would come home and we'd start playing with it, just trying to design things."
Not surprisingly, Geoff enjoyed math and science in school. "It was probably around eighth or ninth grade that I realized math and chemistry were a good fit for me," he says.
One of his strongest memories from this time is learning about molecules. "I remember learning that you can predict the shape of a molecule from where the electrons are, and then learning the properties of the molecule and realizing that this can all be described mathematically," he says. "I started to realize that things are a certain shape for a reason, and that influences how they react. It really brings together all aspects of chemistry."
At the University of Cambridge, Geoff felt fortunate to learn from strong physical chemistry professors who were also very approachable. The discipline of atmospheric chemistry was still in its infancy in the 1970s, but several of his professors dabbled in the area, as issues such as air quality and stratospheric ozone depletion related to the flight of the Concorde supersonic jet began to bring more attention to the field.
Geoff stayed at Cambridge for his Ph.D. in physical chemistry, focusing his research on peroxy radicals that form naturally in the atmosphere and recombine to make hydrogen peroxide. His next move was to Germany for a postdoctoral appointment at the Max Planck Institute. While there, he met scientist A.R. Ravishankara, who recruited him for a postdoctoral position in the Aeronomy Lab at the National Oceanic and Atmospheric Administration in Boulder. In 1988, he came to NCAR.
Understanding 21st century air pollution
Geoff enjoys working in a research setting because of the variety of expertise on staff and opportunities for collaboration. "The measurement people and modeling people are all in the same building, so you can just walk down the hall and find an expert on whatever you want to talk about," he says.
On the horizon, Geoff foresees the kinetics lab conducting more research in support of projects involving air quality and emissions in megacities (extremely large urban areas). He expects gas-phase aerosol interactions to be a major research topic in the future. "It's a challenging field, and there are still plenty of opportunities for young minds to take a look at it," he says.
by Nicole Gordon
April 2009