Methane
Methane is gas that is found in small quantities in the atmosphere. Methane is the simplest hydrocarbon, consisting of one carbon atom and four hydrogen atoms. Methane is a powerful greenhouse gas.
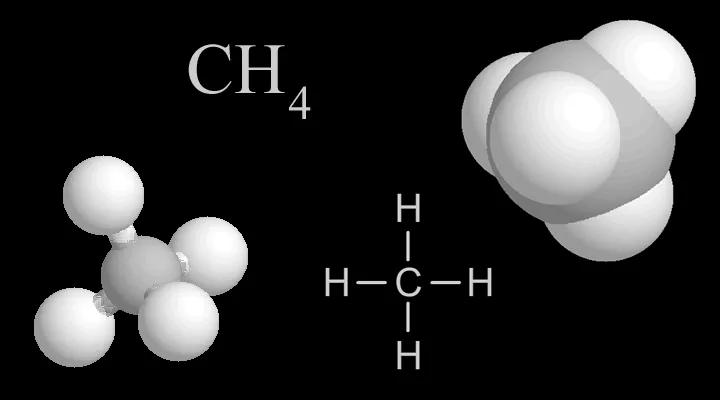
Shown here are four representations chemists use for methane. In the colored models, carbon is light gray and hydrogen is white.
UCAR
Methane is flammable, and is used as a fuel worldwide. It is a principal component of natural gas. Burning methane in the presence of oxygen releases carbon dioxide and water vapor:
CH4 + 2 O2 → CO2 + 2 H2O
Although the concentration of methane in Earth's atmosphere is small (around 1.8 parts per million), it is an important greenhouse gas because it is such a potent heat absorber. The concentration of methane in our atmosphere has risen by about 150% since 1750, apparently largely due to human activities. Methane accounts for about 20% of the heating effects by all of the greenhouse gases combined. Both natural and human sources supply methane to Earth's atmosphere.
Major natural sources of methane include emissions from wetlands and oceans, and from the digestive processes of termites. Sources related to human activities include rice production, landfills, raising cattle and other ruminant animals (cow burps!), and energy generation.
© 2012 UCAR with portions adapted from Windows to the Universe (© 2006 NESTA)