Biogeochemical Cycles
There are a few types of atoms that can be a part of a plant one day, an animal the next day, and then travel downstream as a part of a river’s water the following day. These atoms can be a part of both living things like plants and animals, as well as non-living things like water, air, and even rocks. The same atoms are recycled over and over in different parts of the Earth. This type of cycle of atoms between living and non-living things is known as a biogeochemical cycle.
All of the atoms that are building blocks of living things are a part of biogeochemical cycles. The most common of these are the carbon and nitrogen cycles.
Tiny atoms of carbon and nitrogen are able to move around the planet through these cycles. For example, an atom of carbon is absorbed from the air into the ocean water where it is used by little floating plankton doing photosynthesis to get the nutrition they need. There is the possibility that this little carbon atom becomes part of the plankton’s skeleton, or a part of the skeleton of the larger animal that eats it, and then part of a sedimentary rock when the living things die and only bones are left behind. Carbon that is a part of rocks and fossil fuels like oil, coal, and natural gas may be held away from the rest of the carbon cycle for a long time. These long-term storage places are called “sinks”. When fossil fuels are burned, carbon that had been underground is sent into the air as carbon dioxide, a greenhouse gas.
Recently, people have been causing these biogeochemical cycles to change. When we cut down forests, make more factories, and drive more cars that burn fossil fuels, the way that carbon and nitrogen move around the Earth changes. These changes add more greenhouse gases in our atmosphere and this causes climate change.
The Carbon Cycle
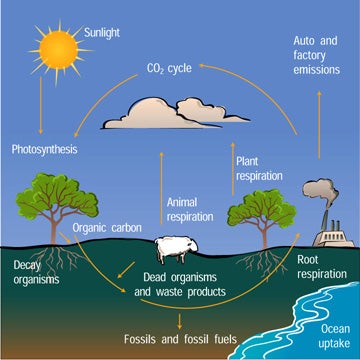
The element carbon is a part of seawater, the atmosphere, rocks such as limestone and coal, soils, as well as all living things. On our dynamic planet, carbon is able to move from one of these realms to another as a part of the carbon cycle.
- Carbon moves from the atmosphere to plants. In the atmosphere, carbon is attached to oxygen in a gas called carbon dioxide (CO2). Through the process of photosynthesis, carbon dioxide is pulled from the air to produce food made from carbon for plant growth.
- Carbon moves from plants to animals. Through food chains, the carbon that is in plants moves to the animals that eat them. Animals that eat other animals get the carbon from their food too.
- Carbon moves from plants and animals to soils. When plants and animals die, their bodies, wood and leaves decays bringing the carbon into the ground. Some is buried and will become fossil fuels in millions and millions of years.
- Carbon moves from living things to the atmosphere. Each time you exhale, you are releasing carbon dioxide gas (CO2) into the atmosphere. Animals and plants need to get rid of carbon dioxide gas through a process called respiration.
- Carbon moves from fossil fuels to the atmosphere when fuels are burned. When humans burn fossil fuels to power factories, power plants, cars and trucks, most of the carbon quickly enters the atmosphere as carbon dioxide gas. Each year, five and a half billion tons of carbon is released by burning fossil fuels. Of this massive amount, 3.3 billion tons stays in the atmosphere. Most of the remainder becomes dissolved in seawater.
- Carbon moves from the atmosphere to the oceans. The oceans, and other bodies of water, absorb some carbon from the atmosphere. The carbon is dissolved into the water.
Carbon dioxide is a greenhouse gas and traps heat in the atmosphere. Without it and other greenhouse gases, Earth would be a frozen world. But since the start of the Industrial Revolution about 150 years ago humans have burned so much fuel and released so much carbon dioxide into the air that global climate has risen over one degree Fahrenheit. The atmosphere has not held this much carbon for at least 420,000 years according to data from ice cores. The recent increase in amounts of greenhouse gases such as carbon dioxide is having a significant impact on the warming of our planet.
Carbon moves through our planet over longer time scales as well. For example, over millions of years weathering of rocks on land can add carbon to surface water which eventually runs off to the ocean. Over long time scales, carbon is removed from seawater when the shells and bones of marine animals and plankton collect on the sea floor. These shells and bones are made of limestone, which contains carbon. When they are deposited on the sea floor, carbon is stored from the rest of the carbon cycle for some amount of time. The amount of limestone deposited in the ocean depends somewhat on the amount of warm, tropical, shallow oceans on the planet because this is where prolific limestone-producing organisms such as corals live. The carbon can be released back to the atmosphere if the limestone melts or is metamorphosed in a subduction zone.
The Nitrogen Cycle
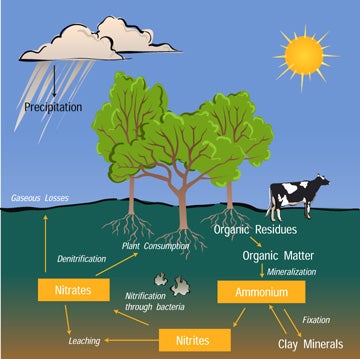
Nitrogen is an element that is found in both the living portion of our planet and the inorganic parts of the Earth system. Nitrogen moves slowly through the cycle and is stored in reservoirs such as the atmosphere, living organisms, soils, and oceans along the way.
Most of the nitrogen on Earth is in the atmosphere. Approximately 80% of the molecules in Earth's atmosphere are made of two nitrogen atoms bonded together (N2). All plants and animals need nitrogen to make amino acids, proteins and DNA, but the nitrogen in the atmosphere is not in a form that they can use. The molecules of nitrogen in the atmosphere can become usable for living things when they are broken apart during lightning strikes or fires, by certain types of bacteria, or by bacteria associated with legume plants. Other plants get the nitrogen they need from the soils or water in which they live mostly in the form of inorganic nitrate (NO3-). Nitrogen is a limiting factor for plant growth. Animals get the nitrogen they need by consuming plants or other animals that contain organic molecules composed partially of nitrogen. When organisms die, their bodies decompose bringing the nitrogen into soil on land or into the oceans. As dead plants and animals decompose, nitrogen is converted into inorganic forms such as ammonium salts (NH4+) by a process called mineralization. The ammonium salts are absorbed onto clay in the soil and then chemically altered by bacteria into nitrite (NO2-) and then nitrate (NO3-). Nitrate is the form commonly used by plants. It is easily dissolved in water and leached from the soil system. Dissolved nitrate can be returned to the atmosphere by certain bacteria through a process called denitrification.
Certain actions of humans are causing changes to the nitrogen cycle and the amount of nitrogen that is stored in reservoirs. The use of nitrogen-rich fertilizers can cause nutrient loading in nearby waterways as nitrates from the fertilizer wash into streams and ponds. The increased nitrate levels cause plants to grow rapidly until they use up the nitrate supply and die. The number of herbivores will increase when the plant supply increases and then the herbivores are left without a food source when the plants die. In this way, changes in nutrient supply will affect the entire food chain. Additionally, humans are altering the nitrogen cycle by burning fossil fuels and forests, which releases various solid forms of nitrogen. Farming also affects the nitrogen cycle. The waste associated with livestock farming releases a large amount of nitrogen into soil and water. In the same way, sewage waste adds nitrogen to soils and water.
Nitrogen and Air Pollution

An unsightly haze of smog, visible from NCAR's Mesa Laboratory, rests over Boulder Valley.
UCAR
Nitric oxide (NO) and nitrogen dioxide (NO2) are together known as nitrogen oxides. These nitrogen oxides contribute to the problem of air pollution, playing roles in the formation of both smog and acid rain. They are released into Earth's atmosphere by both natural and human-generated sources.
Nitric oxide is a colorless, flammable gas with a slight odor. Nitrogen dioxide is a deep red-orange gas that is poisonous but not flammable. It, along with aerosols, is responsible for the reddish-brown color of smog. At high concentrations, it is highly toxic and can cause serious lung damage. Nitrogen dioxide is a strong oxidizing agent, and is thus very reactive with other compounds.
Scientists estimate that between 20 and 90 million tons of nitrogen oxides in produced naturally each year from sources such as volcanoes, oceans, biological decay, and lightning strikes. Human activities add another 24 million tons of nitrogen oxides to our atmosphere annually.
Both NO and NO2 are formed during high-temperature combustion in the atmosphere, when oxygen combines with nitrogen. The exhaust gases of cars and trucks are major sources of nitrogen oxides, as are the emissions from electrical power generation plants. Automobile exhaust has more NO than NO2, but once the NO is released into the atmosphere it quickly combines with oxygen in the air to form NO2.
Nitrogen oxides are at least partially responsible for several types of air pollution. Nitrogen dioxide lends its color to the reddish-brown haze we call smog. Photodissociation of nitrogen dioxide by sunlight produces nitric oxide and ozone in the troposphere, which is another component of smog. A series of chemical reactions transform Volatile Organic Compounds (VOCs) into substances that combine with nitrogen dioxide to produce PAN (Peroxyacytyl nitrate), yet another element in smog. Nitrogen dioxide in the air also reacts with water vapor to form nitric acid, one of the types of acid in acid rain. Nitric oxide concentration in unpolluted air is around 0.01 ppm. In smog, the concentration rises twenty-fold to about 0.2 ppm.
Although nitrogen oxides have gained dubious distinction as pollutants, they are also used beneficially in some industrial processes. Nitric oxide is manufactured on a large scale, and is subsequently used to make nitric acid (HNO3). To create nitric oxide for industrial uses, chemists combine ammonia (NH3) with oxygen (O2), releasing water (H2O) as a byproduct. Nitrogen compounds derived from nitric acid are used to create chemical fertilizers, explosives, and other useful substances.
© 2011 NESTA with modifications by UCAR